8 сар . 26, 2025 04:40 Back to list
Essential Veterinary Injection List: Medicines & Antibiotics
Industry Trends and the Evolving Role of Veterinary Injections
The global animal health market is experiencing robust growth, driven by increasing demand for animal protein, rising pet ownership, and a heightened focus on disease prevention and control. Within this dynamic landscape, the precise and effective administration of medication is paramount, making the veterinary injection list a critical component of modern animal husbandry and veterinary practice. Trends indicate a shift towards advanced formulations, improved drug delivery systems, and a greater emphasis on targeted therapies to minimize resistance and enhance animal welfare.
Stakeholders, including livestock producers, aquaculture farmers, and companion animal veterinarians, increasingly seek high-quality, reliable, and efficacious injectable solutions. The demand extends beyond conventional antibiotics to a broader spectrum of therapeutics, including antiparasitics, vaccines, and nutraceuticals, highlighting the complexity and diversity of the **veterinary injection list** required to address a multitude of health challenges. Data from industry reports suggests the global veterinary pharmaceuticals market is projected to reach over USD 60 billion by 2027, with injectables forming a significant segment due to their rapid action and bioavailability.
Detailed Process Flow for Veterinary Injection Manufacturing
The manufacturing of pharmaceutical-grade veterinary injections is a complex, multi-stage process governed by stringent regulatory standards such as Good Manufacturing Practices (GMP). This ensures the safety, efficacy, and consistent quality of every product on the **veterinary injection list**. The process prioritizes sterility, precision, and active ingredient stability.
1. Raw Material Sourcing and Qualification
This initial phase involves sourcing Active Pharmaceutical Ingredients (APIs) such as Ivermectin and Clorsulon, along with excipients like solvents (e.g., glycerol formal, propylene glycol), antioxidants, stabilizers, and preservatives. All raw materials undergo rigorous quality control inspections, including identity, purity, potency, and heavy metal testing, ensuring compliance with pharmacopoeial standards (USP, EP, BP).
2. Compounding and Mixing
In a controlled environment, precise quantities of APIs and excipients are weighed and dissolved or suspended in sterile water for injection (WFI). This critical step requires specialized mixing equipment to ensure homogeneity and complete dissolution, forming the bulk solution. Temperature and pH are closely monitored to prevent degradation of thermosensitive or pH-sensitive components.
3. Sterilization and Filtration
For injectable formulations, sterility is paramount. The bulk solution typically undergoes sterile filtration through 0.22-micron filters to remove any microbial contamination and particulate matter. This process is performed in an aseptic environment, often a Class 100 (ISO 5) cleanroom, to prevent recontamination. Some formulations may also undergo terminal sterilization, such as autoclaving, if the components are heat-stable.
4. Aseptic Filling and Sealing
The sterile bulk solution is then aseptically filled into pre-sterilized vials or bottles using precision filling machines. These containers are immediately sealed with sterile stoppers and aluminum caps. This process minimizes exposure to the environment, critical for maintaining the product's sterility and preventing contamination that could compromise the efficacy of the **veterinary antibiotic injection list**.
5. Inspection, Labeling, and Packaging
Each filled and sealed vial undergoes a rigorous visual inspection for particulate matter, cracks, or other defects. Automated inspection systems with high-resolution cameras are often employed. Conforming products are then labeled with critical information, including batch number, expiry date, and dosage instructions, before being packaged into cartons for distribution. Stability studies are continuously conducted to determine and confirm the product's service life and shelf-life under various storage conditions.
Schematic Process Flow
Veterinary Injection Manufacturing Process Overview
[Raw Material Reception & QC] ---> [Weighing & Dispensing] --------> [Compounding & Mixing] ----------> [Sterile Filtration] ------------> [Aseptic Filling] ---------------> [Capping & Sealing] -------------> [Visual Inspection] -------------> [Labeling & Packaging] ----------> [Final Quality Control & Release]
This rigorous process ensures that the products are suitable for target industries such as livestock farming (cattle, sheep, swine, poultry), companion animal care, and even aquaculture. Advantages demonstrated include enhanced animal health, reduced disease transmission, and optimized production efficiency, ultimately contributing to the economic viability of farms and the well-being of animals.
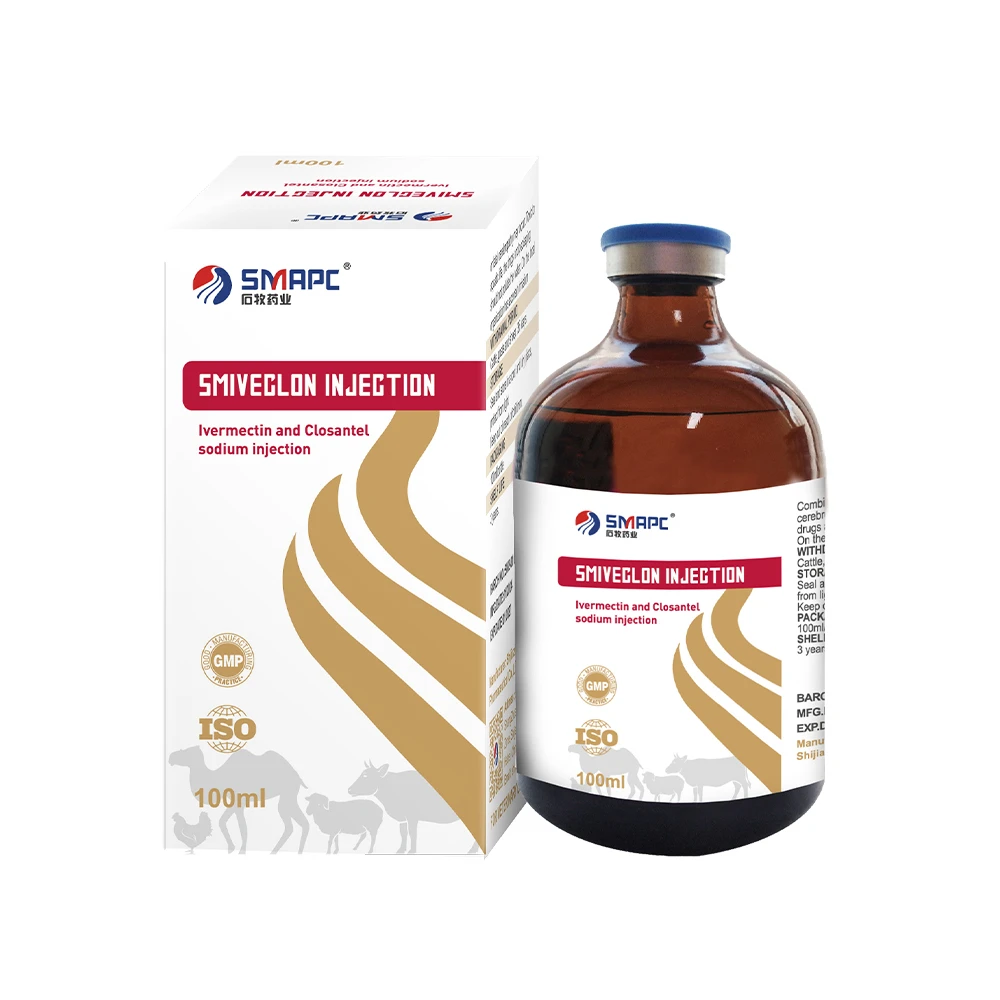
Technical Specifications: Ivermectin and Clorsulon Injection
Focusing on a prominent example from the **veterinary medicine veterinary injection list**, Ivermectin and Clorsulon Injection represents a robust solution for a wide range of parasitic infestations in livestock. Its synergistic formulation combines the broad-spectrum anthelmintic efficacy of Ivermectin with the specific flukicidal action of Clorsulon, targeting both internal and external parasites effectively.
Product Specification Table: Ivermectin and Clorsulon Injection
| Parameter | Specification / Value |
|---|---|
| Active Ingredients | Ivermectin (10 mg/mL), Clorsulon (100 mg/mL) |
| Dosage Form | Sterile Solution for Injection |
| Route of Administration | Subcutaneous (SC) |
| Target Animals | Cattle, Sheep, Goats (as specified by regional regulations) |
| Indications | Treatment and control of gastrointestinal roundworms, lungworms, eyeworms, warbles, mites, lice, and liver fluke. |
| Dosage | 1 mL per 50 kg body weight (for a 0.2 mg/kg Ivermectin and 2 mg/kg Clorsulon dose) |
| Withdrawal Period (Meat) | Typically 28-35 days (varies by region and species) |
| Withdrawal Period (Milk) | Not for use in lactating animals producing milk for human consumption. |
| Storage Conditions | Store below 30°C, protect from light. |
| Shelf Life | 2-3 years unopened, 28 days after first broaching (dependent on specific product data sheet) |
These specifications highlight the critical parameters for effective and safe use. Understanding the pharmacokinetics and pharmacodynamics of each active ingredient, such as the rapid absorption and sustained action of Ivermectin and the targeted hepatic metabolism of Clorsulon, is essential for optimizing treatment protocols and ensuring compliance with regulatory guidelines, particularly concerning withdrawal periods for food-producing animals.
Application Scenarios and Efficacy
The versatility of modern **veterinary injection list** products like Ivermectin and Clorsulon Injection makes them indispensable in various veterinary application scenarios. Primarily targeting ruminants, this combination provides comprehensive parasite control, which is vital for maintaining herd health and productivity.
- Cattle Farming: Used for the routine control of internal parasites (gastrointestinal and lungworms) and external parasites (lice, mites, ticks, warbles). Critical during grazing seasons and for animals entering feedlots. For instance, a single subcutaneous dose can effectively reduce worm burdens, leading to improved weight gain and feed conversion efficiency, crucial metrics for beef production.
- Sheep and Goats: Effective against a similar spectrum of internal nematodes and ectoparasites, including scab mites. Its application helps in preventing chronic diseases that can severely impact wool quality, milk production, and overall animal vigor in small ruminant flocks. Strategic deworming programs integrating such injections have been shown to reduce mortality rates by up to 20% in heavily infested areas.
- Prevention and Control Programs: Integral to proactive herd health management. Administered at strategic times, such as before turnout to pasture or during housing, to break parasite life cycles and minimize environmental contamination. This reduces the need for reactive treatments and can lead to a 10-15% reduction in overall medication costs by preventing widespread outbreaks.
- Disease Eradication Efforts: In regions where specific parasites (e.g., liver fluke) are endemic, targeted injections of products containing Clorsulon are critical for breaking the transmission cycle, particularly where intermediate hosts like snails are prevalent.
The high bioavailability of injectable formulations ensures that therapeutic concentrations of the active ingredients reach the target sites rapidly and sustain their action, leading to quick recovery and long-lasting protection. This directly translates into economic benefits for producers through healthier animals, better growth rates, and reduced veterinary expenses.

Technical Advantages in Modern Veterinary Medicine
The technical advantages of well-formulated **veterinary injection list** products are numerous, offering significant benefits over other administration routes, especially for large animal applications and critical care scenarios. These advantages are rooted in precision, efficacy, and economic efficiency.
- Rapid Onset of Action: Injections, particularly intravenous or intramuscular routes, bypass the digestive system, allowing for quicker absorption into the bloodstream. This rapid systemic distribution is crucial for treating acute infections or severe parasitic burdens where immediate therapeutic levels are required.
- High Bioavailability: Injectable formulations ensure a higher percentage of the active drug reaches systemic circulation compared to oral medications, which can be subject to first-pass metabolism or incomplete absorption. This often allows for lower overall dosages, reducing potential side effects and cost.
- Precise Dosing and Compliance: Injections allow for exact dosing based on animal weight, minimizing under-dosing (leading to resistance) or over-dosing (leading to toxicity). For livestock, administering a single injection can also ensure all animals in a group receive treatment, improving herd compliance compared to feed-based medications.
- Sustained Release Formulations: Advanced injection technologies include long-acting or sustained-release formulations, reducing the frequency of administration. This is particularly valuable in extensive farming systems, cutting down labor costs and minimizing stress to animals.
- Broad-Spectrum Efficacy: Combination products, such as Ivermectin and Clorsulon, offer broad-spectrum control against multiple parasite classes simultaneously, simplifying treatment regimes and offering comprehensive protection.
- Reduced Environmental Impact: Targeted injectable treatments can reduce the overall usage of parasiticides, potentially lowering the environmental burden compared to mass treatments in feed or water, which can contribute to drug residues in the environment.
These technical merits underscore why veterinary injections remain a cornerstone of effective animal health management and contribute significantly to animal welfare and agricultural productivity. The ability to deliver potent therapeutic agents directly and efficiently is a key differentiator.
Vendor Comparison: Key Criteria for Selecting a Supplier
When procuring products for the **veterinary injection list**, selecting the right supplier is as critical as the product itself. B2B decision-makers must evaluate vendors based on a comprehensive set of criteria that ensures product quality, reliability, and long-term partnership value.
Vendor Evaluation Metrics
| Criterion | Description & Importance |
|---|---|
| Regulatory Compliance & Certifications | Ensures adherence to international standards (e.g., GMP, FDA, EMA, ISO 9001). A certified manufacturer guarantees product safety, purity, and efficacy, minimizing legal and health risks. |
| Quality Control & Assurance | Robust internal QC processes, independent batch testing, and comprehensive stability data are crucial. Verifiable test data and CoA (Certificate of Analysis) build trust. |
| R&D Capabilities & Innovation | Suppliers with ongoing R&D can offer advanced formulations, improved drug delivery systems, and solutions for emerging challenges (e.g., antibiotic resistance). |
| Product Range & Availability | A diverse product portfolio covering various species and indications, coupled with consistent supply chain management, is essential for uninterrupted operations. |
| Technical Support & After-Sales Service | Availability of expert veterinary advice, prompt issue resolution, and training support enhances product utilization and overall customer satisfaction. |
| Pricing & Value Proposition | Competitive pricing balanced with superior product quality and service represents optimal value. Total cost of ownership, including efficacy and reduced re-treatment rates, should be considered. |
| Global Reach & Logistics | For international buyers, a supplier's experience in export logistics, cold chain management, and understanding of diverse regulatory environments is critical. |
Prospective partners should conduct thorough due diligence, including site visits, audit reports, and reference checks, to ensure alignment with their operational and ethical standards. A strong vendor relationship built on trust and consistent performance is invaluable in the demanding B2B animal health sector.
Customized Solutions for Specialized Needs
Recognizing that the needs of veterinary practices and large-scale agricultural operations can vary significantly, many leading manufacturers offer customized solutions beyond their standard **veterinary injection list**. This flexibility addresses unique regional disease profiles, animal species, operational scales, and specific regulatory requirements.
- Custom Formulations: Development of specific drug concentrations, combinations of active ingredients, or specialized excipients to improve stability, reduce injection volume, or enhance efficacy for particular conditions. This might involve working with clients to develop a unique **veterinary antibiotic injection list** for a regional bacterial challenge.
- Tailored Packaging and Vial Sizes: Offering different vial sizes (e.g., 50mL, 100mL, 250mL) to match the volume requirements of different farm sizes or veterinary clinics, reducing waste and optimizing logistics. Custom labeling with client branding (private label) is also a common request.
- Regional Regulatory Adaptations: Assisting clients in navigating complex import/export regulations and providing necessary documentation for product registration in specific countries or jurisdictions. This includes modifying formulations or packaging to meet local drug approval standards.
- Specialized Delivery Systems: Investigating novel injection devices or delivery methods that improve ease of use, animal comfort, or drug absorption profiles for specific therapeutic goals.
These bespoke services underscore a manufacturer's commitment to partnership and innovation, providing solutions that are not merely off-the-shelf but are strategically designed to meet the evolving demands of animal health professionals worldwide.
Real-World Application Case Studies
Practical application demonstrates the true value of any product on the **veterinary injection list**. Here are illustrative case studies highlighting the impact of high-quality veterinary injections in various settings:
Case Study 1: Large-Scale Cattle Farm Parasite Control
A commercial beef cattle operation in the Midwestern US faced significant challenges with gastrointestinal nematodes and liver fluke, resulting in reduced weight gain and increased culling rates. After implementing a strategic deworming program using Ivermectin and Clorsulon Injection from a reputable manufacturer, administered twice annually (spring turnout and fall housing), the farm observed remarkable improvements. Fecal egg count reductions averaged 95% for nematodes and 80% for fluke. Average daily gain (ADG) for young stock increased by 10-12% over a single grazing season, translating to an estimated additional USD 50-70 per head at market. This program reduced direct pasture contamination, helping mitigate re-infection pressure, demonstrating the long-term economic benefits.
Case Study 2: Dairy Herd Mastitis Management
A dairy farm with 300 lactating cows experienced a persistent issue with clinical mastitis, primarily caused by *Staphylococcus aureus*. Traditional oral and intramammary treatments yielded inconsistent results. Introducing a new, highly bioavailable veterinary antibiotic injection, as part of a comprehensive treatment protocol for severe cases, significantly improved recovery rates. Cows treated with the injectable antibiotic, alongside supportive therapy, showed a 75% clinical cure rate, compared to 55% with previous treatments. This not only reduced milk discard but also decreased the duration of treatment, leading to a 15% reduction in overall antibiotic usage on the farm over a year, aligning with responsible antibiotic stewardship principles. Customer feedback highlighted the ease of administration and observable rapid improvement in treated animals.
Case Study 3: Swine Production - Respiratory Disease Prevention
A large swine breeding and finishing operation struggled with enzootic pneumonia (Mycoplasma hyopneumoniae) and other respiratory pathogens, particularly in growing pigs. Implementing a preventative vaccination program using a multi-valent injectable vaccine led to a dramatic reduction in respiratory disease incidence. Pre-vaccination, lung lesion scores at slaughter averaged 2.5 (on a 0-4 scale); post-vaccination, this dropped to 0.8. Mortality rates related to respiratory disease decreased by 60%, and average daily feed intake improved by 8%, showcasing the preventative power of a well-chosen **veterinary injection list** item. The use of sterile, high-quality needles and proper injection technique ensured optimal vaccine efficacy and minimal post-injection reactions.

Frequently Asked Questions (FAQ) about Veterinary Injections
Q1: What are the primary advantages of injectable veterinary medications over oral forms?
A: Injectables offer rapid absorption and onset of action, higher bioavailability (meaning more active drug reaches the target), precise dosing, and are often easier to administer to uncooperative animals. They are also essential for drugs that would be degraded by the digestive system.
Q2: How important is proper administration technique for veterinary injections?
A: Extremely important. Correct administration technique ensures drug efficacy, minimizes adverse reactions (e.g., abscesses, nerve damage), reduces animal stress, and adheres to withdrawal periods. Training and adherence to manufacturer guidelines are crucial.
Q3: What are "withdrawal periods" and why are they critical for food-producing animals?
A: Withdrawal periods are the time intervals required between the last drug administration and when the animal's products (meat, milk, eggs) can safely enter the human food chain. They are critical to ensure that drug residues in animal products are below established maximum residue limits (MRLs), protecting public health and ensuring food safety compliance.
Q4: Are there concerns about antimicrobial resistance with veterinary antibiotics?
A: Yes, antimicrobial resistance (AMR) is a significant global concern in both human and animal medicine. Responsible use of the **veterinary antibiotic injection list**, guided by veterinary diagnosis, proper dosing, and adherence to treatment durations, is essential to mitigate AMR development.
Q5: How should veterinary injections be stored to maintain their efficacy?
A: Most injections require storage in a cool, dark place, typically below 30°C, and protected from freezing or direct sunlight. Specific temperature ranges are indicated on product labels. After broaching, certain products have a limited shelf life (e.g., 28 days) and should be used or discarded accordingly.
Lead Time, Warranty, and After-Sales Support
For B2B clients, transparency regarding logistics, product assurances, and ongoing support is paramount. Reputable manufacturers provide clear commitments in these areas to build lasting partnerships.
- Lead Time and Fulfillment: Standard lead times for stock items typically range from 2-4 weeks, depending on order volume and destination. For customized or bulk orders, lead times may extend to 6-12 weeks, with detailed production schedules provided at the time of order confirmation. Global shipping is managed through established logistics partners, ensuring cold chain integrity where required, and efficient customs clearance.
- Warranty Commitments: Products are warranted to meet all specified parameters, potency, sterility, and stability as per their Certificate of Analysis and relevant pharmacopoeial standards (e.g., USP, EP, BP) up to their stated expiry date, provided they are stored and handled according to label instructions. Any product failing to meet these quality standards will be subject to replacement or credit, following investigation.
- Customer Support and Technical Assistance: Comprehensive after-sales support is offered, including a dedicated technical service team comprising veterinary professionals. This team provides guidance on product selection, optimal usage protocols, dosage adjustments, and troubleshooting. Support is available via phone, email, and online portals, ensuring prompt resolution of inquiries and continuous client education.
This robust framework of logistics, quality assurance, and support services ensures that clients receive not just products, but complete solutions that contribute to their operational success and animal health outcomes.

Conclusion
The field of veterinary pharmaceuticals, particularly the development and application of the **veterinary injection list**, is indispensable for modern animal health management. From rigorous manufacturing processes adhering to global GMP standards to the strategic application of advanced formulations like Ivermectin and Clorsulon Injection, these products are instrumental in preventing disease, improving animal welfare, and enhancing agricultural productivity. As the industry continues to evolve, driven by technological innovation and a commitment to animal health and food safety, the importance of high-quality, scientifically-backed injectable solutions will only grow. Partnering with reputable manufacturers who prioritize quality, innovation, and comprehensive support is key to navigating the complexities of animal health and achieving sustainable success in the B2B sector.
References
- OIE - World Organisation for Animal Health. (2022). OIE Terrestrial Animal Health Code. Retrieved from https://www.oie.int/en/what-we-do/standards/codes/terrestrial-code/
- EMA - European Medicines Agency. (2021). Veterinary Medicines. Retrieved from https://www.ema.europa.eu/en/veterinary-medicines
- FDA - U.S. Food and Drug Administration. (2023). Veterinary Drugs. Retrieved from https://www.fda.gov/animal-veterinary
- USP - United States Pharmacopeia. (2023). Veterinary Drug Products. Retrieved from https://www.usp.org/
- Grand View Research. (2020). Veterinary Pharmaceuticals Market Size, Share & Trends Analysis Report. Retrieved from https://www.grandviewresearch.com/industry-analysis/veterinary-pharmaceuticals-market
-
Premium Vet Powder for Dogs: Antibacterial & Calcium SupportNewsAug.29,2025
-
Veterinary Calcium Powder: Bone Health & Growth SupportNewsAug.28,2025
-
Polyhexamethylene Biguanide Hydrochloride Role in Modern DisinfectionNewsAug.21,2025
-
Polyhexamethylene Biguanide Hydrochloride: Chemical Definition and Industrial SignificanceNewsAug.21,2025
-
Essential Storage & Handling for OTC Expectorant SolutionsNewsAug.21,2025
-
Decoding Expectorant Strength in Multi-Symptom Cold Expectorant FormulasNewsAug.21,2025
Products categories