Oct . 12, 2025 13:35 Back to list
Veterinary Injection List | GMP-Certified, Dosage Guides
A field insider’s take on the veterinary injection list and why Compound Vitamin B matters
If you work barns, feedlots, or small animal clinics, you already know: the veterinary injection list is crowded—antimicrobials, NSAIDs, biologics, electrolytes, trace minerals, and, yes, vitamin complexes. Trends? Actually pretty clear. Fewer products that try to do everything, more targeted support, cleaner formulations, and better QC. One item I keep seeing in procurement notes is Compound Vitamin B Injection—simple, practical, and (to be honest) a quiet workhorse when appetite and metabolism wobble.
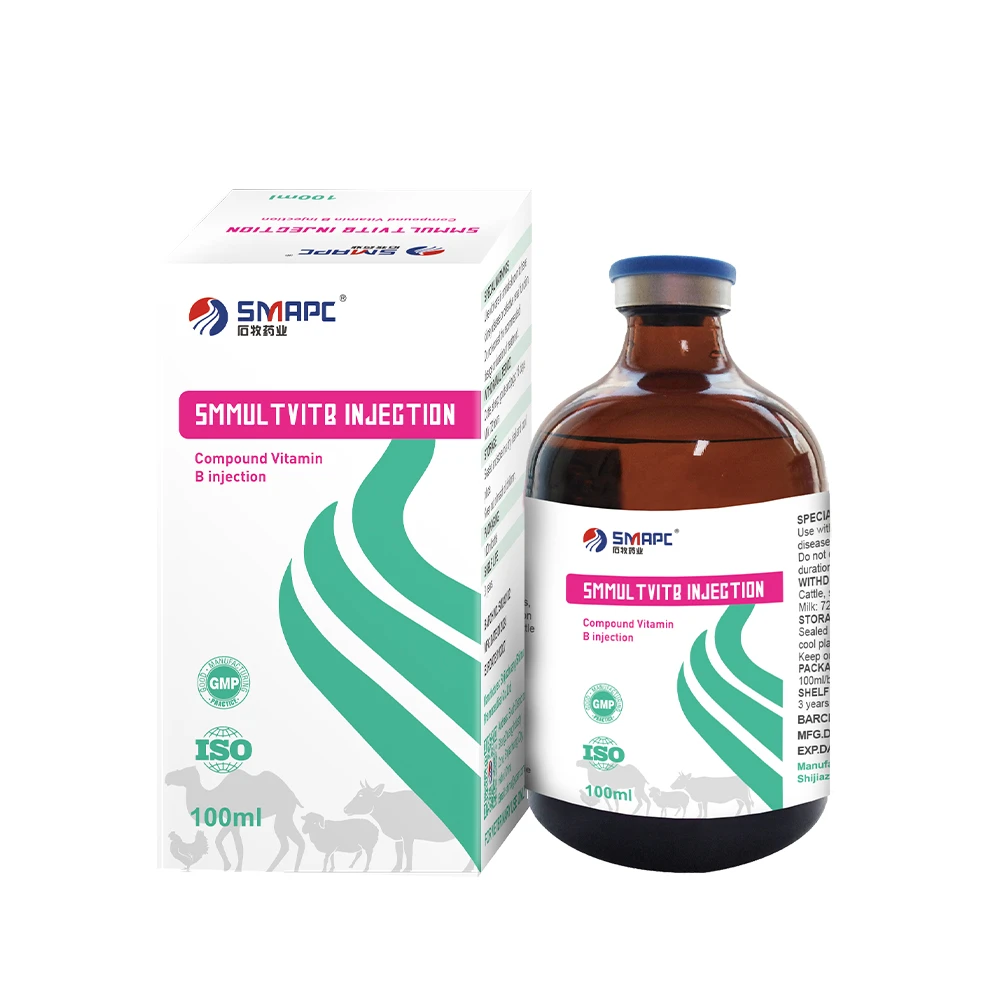
Product snapshot: Compound Vitamin B Injection
Factory origin: South District of Shangzhuang Industry Zone, Shijiazhuang, Hebei, China. Many customers say it’s a “reach-for-it-first” when animals show stress-related inappetence or nervous signs.
| Parameter | Specification (≈ real-world use may vary) |
|---|---|
| Composition (per ml) | VB1 10 mg; VB6 1 mg; VB2 1 mg; Nicotinamide 15 mg; Sodium D-pantothenate 0.5 mg |
| Appearance | Clear, sterile solution |
| Indications | Support in multiple neuritis, digestive disorders, ecdysis issues, stomatitis due to B-vitamin deficiency |
| pH / Osmolality | pH 5.0–6.5 (typ.); isotonic-adjusted vehicle |
| Packaging | Glass vials or bottles (e.g., 10 ml, 50 ml, 100 ml) |
| Shelf life | ≈ 24 months sealed; store below 30°C, protect from light |
Process flow and quality gates (how it’s made, briefly)
- Materials: pharma-grade thiamine, riboflavin, pyridoxine, nicotinamide, sodium D‑pantothenate; WFI-grade solvent; stabilizers as needed.
- Methods: controlled weighing → sequential dissolution → pH/osmolality tuning → 0.22 μm sterile filtration → nitrogen flush → aseptic filling → stoppering/capping → visual inspection.
- Testing standards: Sterility per USP <71>; Endotoxins per USP <85>; Particulates per USP <788>; Assay by HPLC (98–102% label claim typical); Container-closure integrity per USP <1207>; Stability per VICH/ICH guidance.
- Service life: validated by accelerated/long-term studies (e.g., 25°C/60% RH and 30°C/65% RH profiles).
- Industries: dairy and beef operations, swine integrators, small ruminants, equine, and mixed-practice clinics.
Where it fits in the veterinary injection list
Use during transport stress, post-antibiotic recovery, poor thrift, or feed transitions. As one Hebei dairy manager told me, “It’s the small boost that gets appetite back on track.” Not magic—just solid cofactor support for energy metabolism.
Typical batch-release data (illustrative)
- Assay: VB1 100.6%, VB2 99.2%, VB6 101.1%, Nicotinamide 99.8%, Pantothenate 98.7% (HPLC)
- Endotoxins: < 0.5 EU/ml; Sterility: Pass (14-day)
- Particulate: Within USP <788> small-volume injection limits
Vendor comparison (what buyers usually ask)
| Vendor | GMP/ISO | MOQ | Lead Time | Private Label | Notes |
|---|---|---|---|---|---|
| Skyvet Pharm (Hebei factory) | Veterinary GMP; ISO 9001 | ≈ 5,000–10,000 vials | 3–5 weeks | Yes (artwork + stability files) | Factory-direct QC and pricing |
| Vendor A (regional) | ISO 9001 | ≈ 2,000 vials | 4–6 weeks | Limited | Broker-based sourcing |
| Vendor B (export-focused) | Veterinary GMP | ≈ 10,000 vials | 5–7 weeks | Yes | Requires long-term contract |
Customization and documentation
Common tweaks: vial sizes, label languages, carton specs, COA formats, and region-specific registration dossiers. Stability data files and method validations are often shared under NDA—worth asking early.
Quick case note
A mixed-practice clinic reported faster feed return in stressed weaners when Compound Vitamin B was paired with fluids and probiotics. Small sample, sure, but the feedback repeats across buyers: “simple support that plays well with others.”
Regulatory reminder: follow local veterinary guidance, product labeling, and withdrawal stipulations where applicable.
Authoritative citations
- USP <71> Sterility Tests; USP <85> Bacterial Endotoxins Test; USP <788> Particulate Matter in Injections; USP <1207> Container Closure Integrity.
- VICH GL39(R) Stability Testing of New Veterinary Drug Substances and Medicinal Products.
- ISO 9001:2015 Quality Management Systems—Requirements.
- China GMP for Veterinary Drugs (latest revision), National Medical Products Administration (NMPA)/MOA notices.
-
Choosing the Best Worm Medicine for Goats for Herd HealthNewsApr.16,2026
-
Comprehensive Guide to Professional Goat Medication and Herd HealthNewsApr.09,2026
-
Comprehensive Tick Treatment for Sheep A Guide to Control and PreventionNewsApr.07,2026
-
Comprehensive Guide to Effective Mite Treatment for Goats and PreventionNewsApr.04,2026
-
Comprehensive Veterinary Medicine for Sheep Health and Disease PreventionNewsMar.31,2026
-
Comprehensive Guide to Goat Health and Effective Goats Medicine SolutionsNewsMar.28,2026
Products categories







